INTRODUCTION
Microtubule-associated proteins (MAPs) provide critical support to the neuronal cytoskeleton and mediate intracellular transport and migration. Growing evidence suggests that MAPs may be involved in the dynamics underlying dendritic and axonal retraction during neurodegeneration.1 The most infamous MAP isoform, tau, is an established biomarker of dementia and cognitive dysfunction2,3 and has been previously associated with postoperative delirium.4–7 Of the four major families of MAPs, MAP2 has been understudied as a biomarker of neurological conditions but is highly enriched in the brain compared to the periphery8 and serves a critical role in preserving neuronal structure, neurogenesis, and neuroplasticity.9,10 We hypothesize that blood MAP2 levels may reflect dynamic changes occurring in the brain in response to major cardiac surgery and indicate risk of cognitive dysfunction. Here, we developed custom single-molecule immunoassays to detect MAP2 in serum and investigate potential associations with the development of postoperative delirium and neuronal stress.
METHODS
Clinical Study
All studies were approved by the Mass General Brigham Institutional Review Board (Protocol #: 2018P000480 and 2022P000445). Serum samples derived from subjects undergoing major cardiac surgery were analyzed from 2 independent cohorts.11,12 Cohort 1 was composed of 19 subjects with serum collected before surgery and on postoperative day 1. Cohort 2 was composed of 57 subjects with serum collected at 6 perioperative timepoints: immediately before surgery, at the start and end of cardiopulmonary bypass, and on postoperative days 1 – 3. Inclusion criteria for both cohorts included older subjects (≥ 60 years) scheduled for major cardiac surgery with cardiopulmonary bypass and postoperative cardiac ICU admission to permit blood collection. Exclusion criteria included blindness, deafness, and the inability to speak English, renal or liver failure, COVID-symptomatic or positive, > 2 days in the ICU in the month prior to surgery, and severe neurocognitive damage. Delirium was assessed twice daily using the Confusion Assessment Method by trained clinical research staff.13 We have previously published serum analysis of Cohort 16 and Cohort 2.12,14
Biomarker Analysis
The Simoa homebrew technology15 was utilized to generate custom Simoa plates using validated paired antibodies for binding and detection of MAP2 (Supplemental Methods).
SH-SY5Y Study
Differentiated SH-SY5Y cells derived from a neuroblastoma cell line were cultured in vitro and stimulated with acute glutamate to induce excitotoxicity (Supplemental Methods).16
Statistical Analyses
Effect estimates are presented as fold-change or median difference and their associated standard deviation or 95% confidence intervals (CIs). No imputation of missing data was performed, and only datapoints outside of the limits of quantification were excluded. No adjustments for covariates were made due to the small sample sizes and risk of overfitting. To identify possible associations with delirium, the values were log-transformed and normalized to the preoperative MAP2 concentration comparing no delirium and delirium groups using a Mann-Whitney U test. The normalized fold-change in MAP2 concentrations at each timepoint from baseline was assessed with a Wilcoxon matched-pairs signed rank test. Statistical analysis for the MAP2 in vitro studies was based on a Brown-Forsythe and Welch’s ANOVA test. For all analyses, two-sided p-values < 0.05 were considered statistically significant.
RESULTS
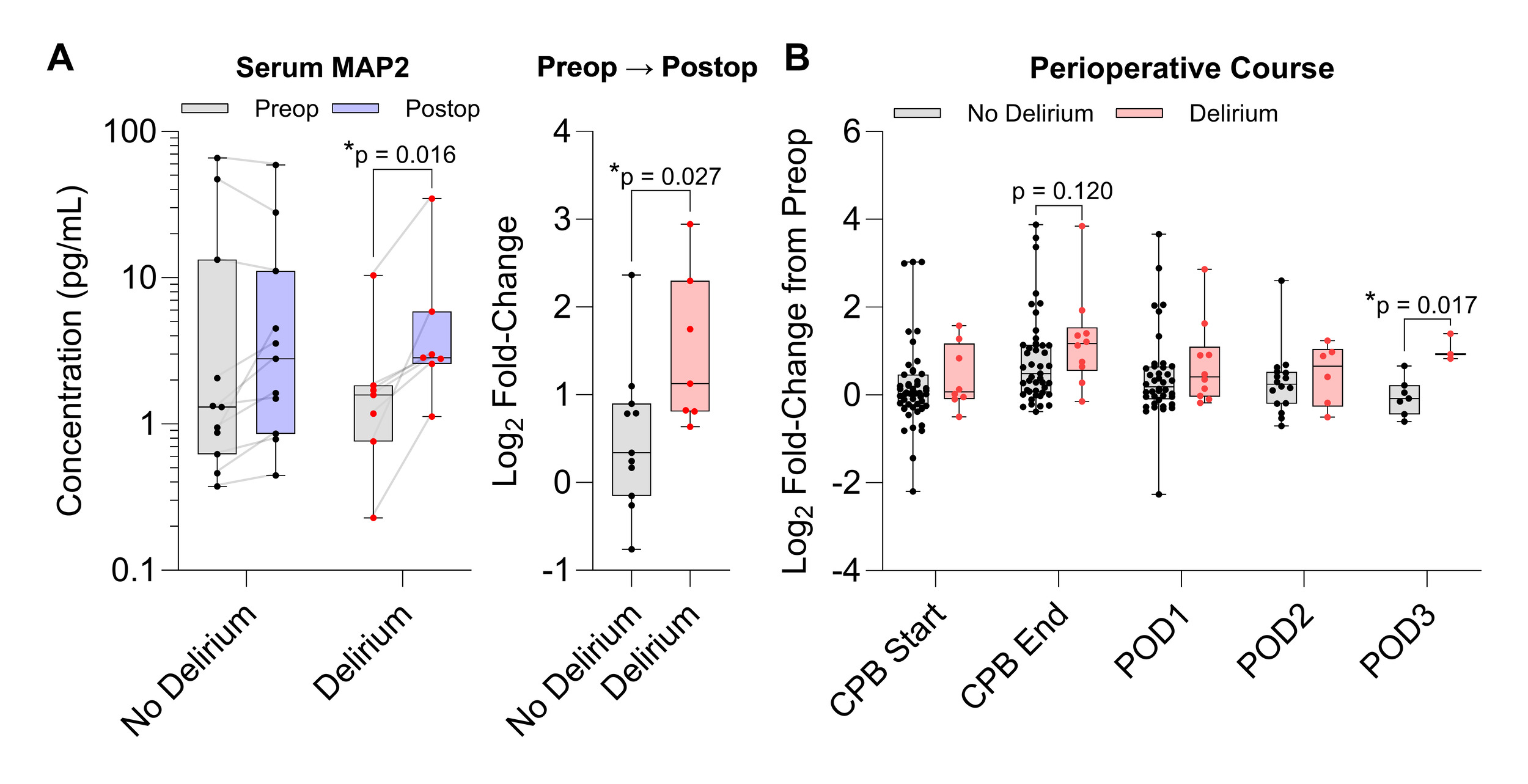
Sera collected from two independent cohorts composed of patients aged 60 years and older undergoing major cardiac surgery with cardiopulmonary bypass at Massachusetts General Hospital were analyzed (Table 1). In general, circulating MAP2 levels increased after surgery by postoperative day 1 compared to baseline (Supplemental Figure 1 – 2). In Cohort 1, patients who experienced postoperative delirium showed significantly higher MAP2 from preoperative values (2.74-fold, 95% CI [1.66 – 5.09], p = 0.016, n = 8) and a larger change to postoperative day 1 (1.94-fold, 95% CI [1.02 – 3.89], p = 0.027) compared to patients who did not develop delirium (n = 11, Figure 1A and Supplemental Table 1). Correlation analysis revealed a positive association between preoperative levels of MAP2 and P-Tau-231, which has been reported as a predictive biomarker of Alzheimer’s disease17 (Supplemental Figure 3). In Cohort 2 with intraoperative blood sampling, MAP2 levels peaked at the end of cardiopulmonary bypass and gradually lowered up to postoperative day 3 in patients who did not develop delirium (Supplemental Figure 4). Subjects who experienced delirium showed a modestly higher increase in MAP2 at the end of cardiopulmonary bypass (1.55-fold, 95% CI [-0.40 – 3.45], p = 0.12, n = 8) and postoperative day 3 (3.59-fold, 95% CI [0.91 – 6.09], p = 0.017, n = 3) relative to preoperative levels compared to patients who did not develop delirium (n = 45 and n = 7, Figure 1B and Supplemental Table 2). In both cohorts, delirium incidence was more strongly associated with the postoperative change in MAP2 from baseline values, rather than preoperative or postoperative timepoints alone, suggesting that the increase in serum MAP2 may reflect a vulnerability to surgical stressors.
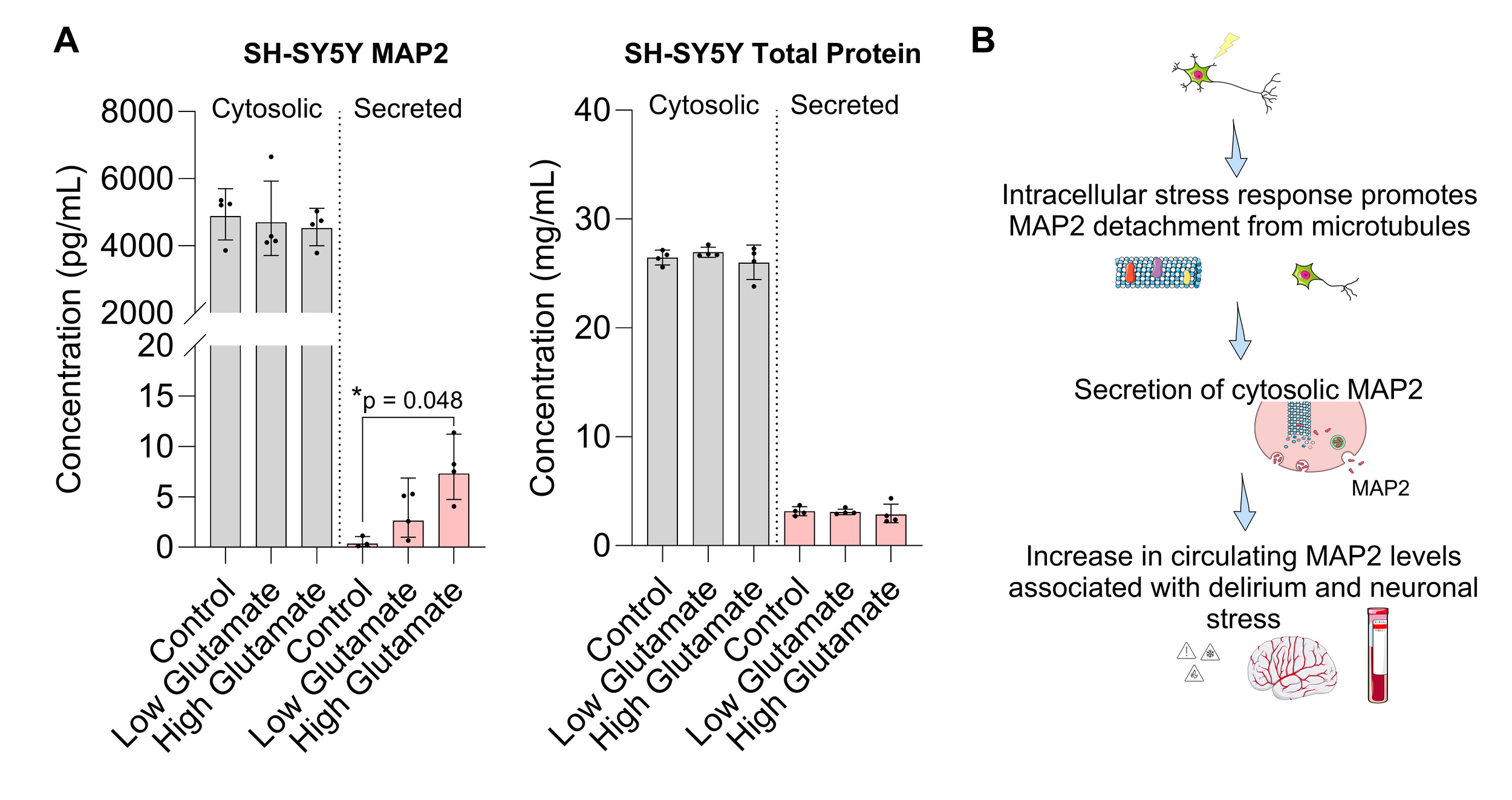
To provide mechanistic insight into extracellular MAP2 as an indicator of neuronal stress, we examined MAP2 secretion using an in vitro model of differentiated SH-SY5Y cells stimulated with glutamate to induce excitotoxicity. Extracellular MAP2 levels in the media significantly increased in a dose-dependent manner following acute glutamate exposure (13.55-fold increase, 95% CI [1.47 – 125.20], p = 0.048) with no significant change in total cytosolic protein levels, suggesting that MAP2 may be selectively secreted in the absence of overt cell lysis (Figure 2A). Our working hypothesis is that surgical stress promotes release of MAP2 from microtubules, leading to dendritic retraction and secretion of unbound, cytosolic MAP2 into the extracellular space and increased circulating levels in blood (Figure 2B). Collectively, our results lend support to further studies evaluating the use of circulating MAP2 as a surrogate marker of acute neurotoxicity and postoperative delirium.
CONCLUSION
Delirium remains a relatively common complication of major cardiac surgery. The discovery and validation of sensitive blood-based biomarkers associated with postoperative delirium may enable the development of targeted interventions to promote cognitive resilience. This study developed and optimized custom assays to quantify the abundance of MAP2 in two independent cohorts of older adults undergoing cardiac surgery and identified possible associations with delirium based on quantitative protein measures. We selected MAP2 as a starting biomarker with unexplored potential given its specificity for the brain and hypothesized that its localization primarily to neuronal dendrites may provide a targeted picture of cognitive changes in response to surgical stress. The multifactorial pathophysiology involved in postoperative delirium has made it difficult to identify a therapeutic approach to improve resilience to surgical stressors. The ability to identify patient susceptibility to delirium using objective biomarkers, such as MAP2, may provide a means to assess whether targeted interventions are effective. Our results show that MAP2 protein levels increase in serum after major cardiac surgery and may be associated with postoperative delirium. The findings support our previous studies evaluating tau and neurofilament light chain as markers of neurodamage and possible associations with delirium.6,12 Longitudinal studies evaluating circulating MAP2 as a predictive marker of long-term cognitive changes may aid in defining its clinical utility in the context of established biomarkers of Alzheimer’s disease and related dementias. Understanding how changes in the blood-brain barrier may influence serum MAP2 levels and their association with cognitive outcomes may also be important areas to explore. The small samples sizes in our study are a notable limitation. Further studies validating these findings in larger, more diverse cohorts are required to adjust for confounders including age, sex, and baseline cognition. This short research report provides evidence that MAP2 may be a novel biomarker of postoperative delirium and is released from neuronal-like cells upon exposure to glutamate highlighting the need to explore the MAP family beyond tau protein in the study of delirium.
Acknowledgments
We would like to acknowledge the Network for Investigation of Delirium (NIDUS) for a seed award (TBM). Pictorials for the schematic shown in Figure 2B were modified from Servier Medical Art available under a Creative Commons license (CC BY 4.0).
Authors’ Contributions
Conception: TBM, OA
Design: TBM, OKG, AT
Data acquisition: TBM, OKG, AT
Data analysis: TBM, OKG, AT
Data interpretation: TBM, OKG, AT, CS, OA
Drafting/revising/approving of final paper: TBM, OKG, AT, CS, OA
Ethics Statement
This study was approved by the Mass General Brigham Institutional Review Board (Protocol #: 2024P002698).
Funding Sources
This work was supported by the National Institutes of Health (R21AG073750, R01AG083777, and R33AG071744).
Declarations of Interests
OA is a consultant and holds equity in Reversal Therapeutics.